Pathogenesis
Pathogenesis information of NAFLD is manually extracted from research articles.
Disease pathway of NAFLD (Kyoto Encyclopedia of Genes and Genomes)
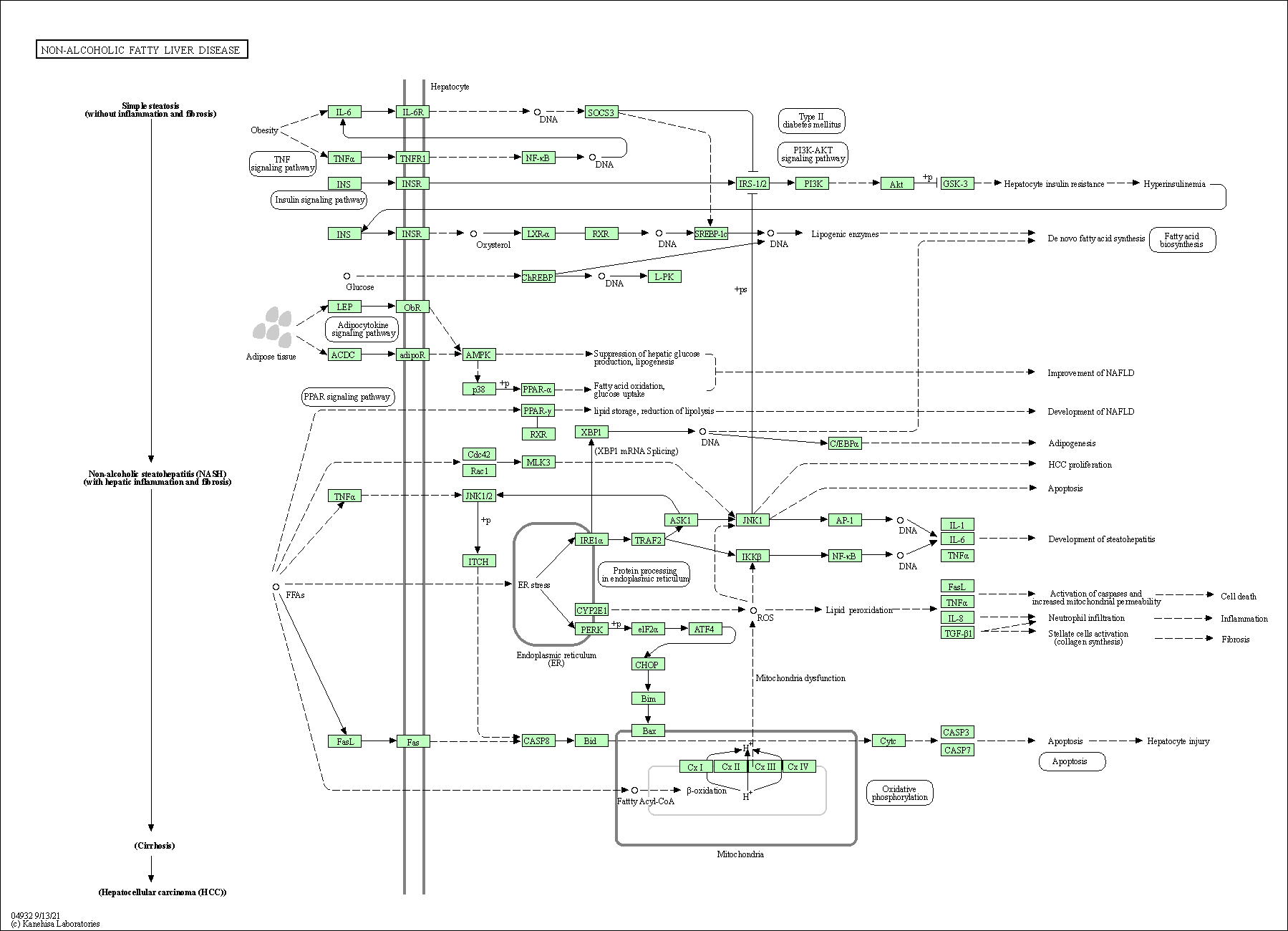
8 records.
| Path ID | Pathogenesis | Other Terms | Description | Reference (PMIDs) |
|---|---|---|---|---|
| P01 | Genetic variant | Gene variant; Heredity; Genetic factor; Epigenetic modifications | Human genetic analyses have provided evidence for some degree of heritability of NAFLD, which could at least partly explain the broad variability in phenotypes and risk of disease progression; Genetic variants, especially in the form of single nucleotide polymorphisms, influence hepatic FFAs flux, oxidative stress, response to endotoxins and cytokine production and activity, and are determinants of NAFLD development and progression. | 33220492; 26823198; 21538280; 26299412; 32910776; 24927523 |
| P02 | Dietary factor | Nutrition; Excessive caloric intake; Specific nutrients; Hypercaloric diet; High-fat diet; Environmental factors | Excessive caloric intake, a nutritional pattern rich in saturated fat, carbohydrates, and sugar-sweetened beverages have all been implicated in the development of NAFLD; Dietary factors, both in terms of quantity and caloric intake but also of specific nutrients, contribute to the development of NAFLD and NASH. | 33220492; 26823198; 18276725; 28112681; 32434996 |
| P03 | Gut microbiota factor | Gut microbiome; Intestinal microbiota; Microbiota factor; Gut–Liver axis | The gut microbiome is implicated in the pathogenesis and progression of NAFLD, through the so-called gut-liver axis; The body of evidence supporting an association between gut microbiome and NAFLD is increasing, as illustrated by a surprisingly robust diagnostic accuracy of gut microbiota-derived signature for predicting the presence of advanced fibrosis F3 or F4 in 86 well-characterized individuals with biopsy-proven NAFLD. | 33220492; 26823198; 31484056; 19291785; 22972297 |
| P04 | Adipose tissue dysfunction | Adipose tissue metabolic dysfunction; Adipose–Liver axis; Obesity | Adipose tissue metabolic dysfunction is closely related to liver inflammation and fibrosis in humans and is a central driver of NASH development; Adipose tissue contributes to the maintenance of low-grade inflammatory states by producing pro-inflammatory cytokines, increased expression of inflammatory genes and macrophages activation in the visceral and subcutaneous adipose tissue of patients with NAFLD correlates with progression from simple steatosis to NASH and fibrosis. | 32258944; 33220492; 26823198; 26028579 |
| P05 | Hepatic lipid accumulation | Fat metabolism; Lipotoxicity; Lipid accumulation; Lipid metabolism; Hepatocyte insulin resistance; Endoplasmic Reticulum Stress; Mitochondrial Dysfunction | Net hepatic TAG accumulation is the result of hepatic lipid supply and demand. The FA transporters (FATP2 and FATP5) and the cluster of differentiation 36 (CD36) facilitate hepatocyte FFA uptake. Studies have reported conflicting findings on the expression patterns of FATP2 and CD36 in humans with steatosis and NASH. An inverse association between hepatic FATP5 messenger RNA expression and histologic changes in NAFLD, including ballooning and fibrosis, suggesting reduced FATP5-mediated TAG accumulation during NASH progression; Increased triglyceride concentration is an epiphenomenon which happens simultaneously with toxic metabolites generation, lipotoxicity and liver injury; DNL, namely, the conversion of acetyl-CoA over malonyl-CoA via acetyl coenzyme A carboxylase (ACC) and finally to palmitate, was independently associated with hepatic TAG in individuals with NAFLD. | 33220492; 26823198; 31602526; 24316260; 18072723 |
| P06 | Intrahepatic inflammation | Immune dysregulation; Inflammasome activation | The activation of inflammasomes in response to FFAs, oxidative stress and other pro-inflammatory metabolites seen in NAFLD with consequent production of IL-1β could have an important role in the development of NASH, by inducing suppression of peroxisome proliferator-activated receptor-α (PPAR-α), and promoting the indirect effect of TNF-α-induced cell death. | 33220492; 26823198; 30482910; |
| P07 | Intrahepatic fibrosis | Hepatic fibrosis; Liver fibrosis | Fibrosis is the result of excessive production of extracellular matrix (ECM) that is not adequately balanced by degradation, thus resulting in net accumulation. In the liver, hepatic stellate cells (HSCs) constitute the main source of ECM-producing fibroblasts in models of toxic and biliary liver disease and NAFLD. | 33220492; 32085494; 32044315 |
| P08 | Metabolic comorbidity | Metabolic syndrome; Metabolic abnormality; Complications | NAFLD is mainly affecting individuals with obesity and type 2 diabetes mellitus (T2DM). Despite the general association among obesity, dysglycemia, and NAFLD, the presence of metabolic comorbidities coexisting with NAFLD varies substantially across populations; By contrast, obesity cannot be regarded as the sole criterion for the screening of NAFLD, because the prevalence of nonobese NAFLD varies in the general population from 25% to 50% and the presence of ≥1 metabolic abnormalities even in the absence of obesity doubles the risk for NAFLD. | 33220492; 32605642; 32413340; 16932311 |
